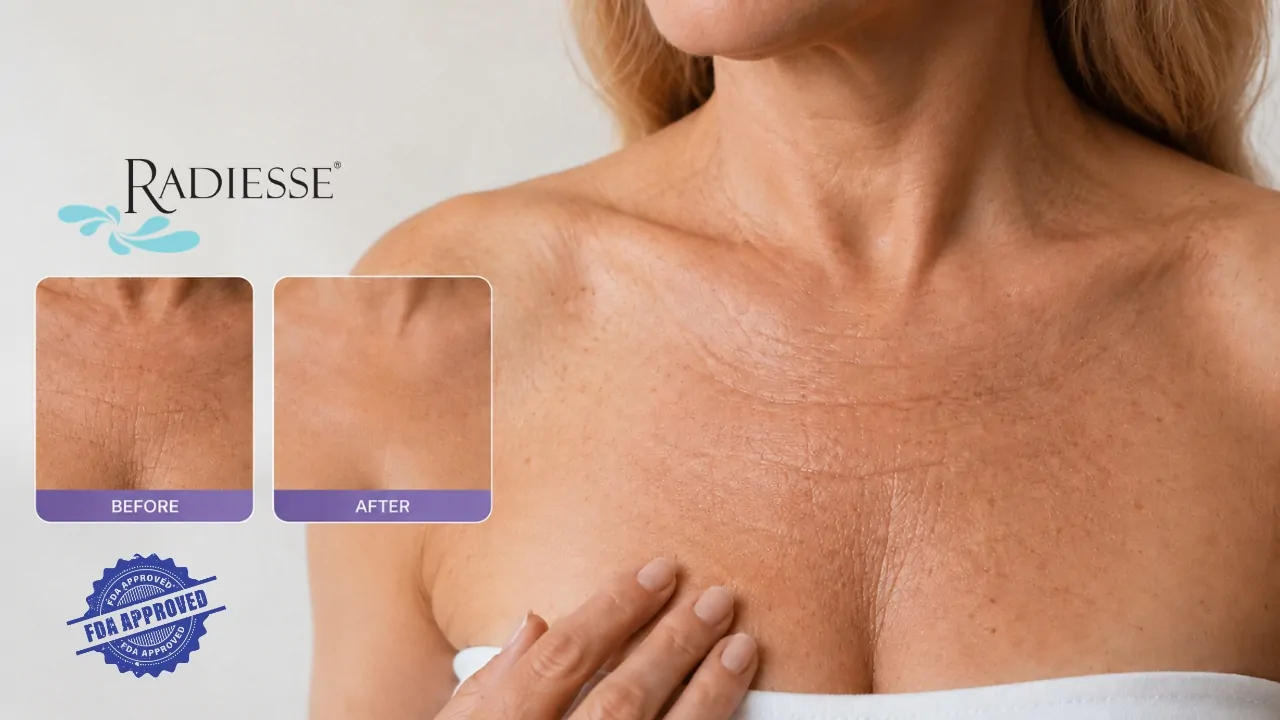
Radiesse has gained a new FDA-approved use for treating wrinkles in the upper chest, also called the décolleté, giving aesthetic providers a cleared injectable option for an area that often ages faster than patients expect.
The approval matters because chest wrinkles have long been treated with lasers, peels, energy devices, skin care, or off-label injectables, while the new indication gives Radiesse a specific regulatory pathway for adults 22 and older.
Table of Contents
ToggleWhat The FDA Approved
The FDA approved Radiesse Injectable Implant diluted 1:2 with 0.9% sterile saline for subdermal implantation to correct décolleté wrinkles in patients 22 years of age and older. The agency’s PMA supplement record lists the decision date as March 31, 2026, under supplement P050052/S162.
For the décolleté indication, Radiesse is not used in the same thick, volume-building manner many people associate with facial fillers. FDA labeling says the product should be diluted with sterile saline at a 1:2 ratio and injected with a 22-gauge flexible cannula.
That detail matters. The upper chest is thin-skinned, mobile, sun-exposed, and close to breast tissue. The approved approach focuses on improving wrinkling across a defined central chest area rather than adding obvious volume.
Quick Look At The New Indication
Detail
FDA-Cleared Information
Product
Radiesse Injectable Implant
New treatment area
Décolleté, or upper chest
Approved patient group
Patients 22 years and older
Dilution
1:2 with 0.9% sterile saline
Injection plane
Subdermal implantation
Tool listed in labeling
22-gauge flexible cannula
Approval decision date
March 31, 2026
Why Upper Chest Wrinkles Are Hard To Treat

The upper chest ages in a slightly different way than the cheeks or lips. Wrinkles often come from repeated folding during side sleeping, sun exposure, thinner skin, and gradual loss of collagen and elasticity.
Instead of one deep crease, many patients develop a crinkled texture, vertical sleep lines, or a mismatch between a smoother face and a more weathered chest.
That mismatch has become more visible as facial treatments have improved. Someone may use sunscreen, retinoids, neuromodulators, fillers, or lasers on the face while the chest receives less attention.
By the time décolleté wrinkles become obvious, topical products alone usually cannot create the same level of change expected from an in-office procedure.
The FDA’s summary of safety and effectiveness also lists several alternative approaches for décolleté aging, including neurotoxins, other dermal fillers, chemical peels, intense pulsed light, lasers, and radiofrequency or ultrasound skin tightening.
The same FDA document notes that each option carries different advantages and limitations, so selection should happen through a physician consultation.
How Radiesse Works In Simple Terms
Radiesse is made with synthetic calcium hydroxylapatite, often shortened to CaHA, suspended in a gel carrier. FDA labeling describes CaHA particles in the 25 to 45 micron range.
Because Radiesse sits in a different category from hyaluronic acid fillers, provider-side product education and sourcing matter. Ogomed.com lists Radiesse among its professional dermal filler options for qualified medical buyers.
In aesthetic use, Radiesse has long been used for facial wrinkles and folds, HIV-associated facial lipoatrophy, and hand volume loss before the new décolleté expansion.
In the chest, dilution changes the treatment goal. The diluted product spreads more broadly across the thin tissue layer, supporting a smoother look over time. A patient should not expect the same effect as a thick filler placed in the cheekbone or jawline.
Merz Aesthetics describes Radiesse as a regenerative biostimulator designed to support collagen, elastin, and other skin-structure proteins, with company statements saying results can last up to 2 years.
That duration claim should be read alongside the FDA clinical data, which followed treated patients out to 84 weeks in the pivotal study.
The Clinical Study Behind The Approval
Big news for our skin goals! Radiesse is officially FDA-approved for the décolleté. Stop neglecting your chest area and let’s keep that skin looking smooth, plump, and youthful. ?✨ #skincare #radiesse #antiaging #glowup #dermatology pic.twitter.com/wRySlOaLhP
— Aire MedSpa (@AireMedspa) April 29, 2026
The FDA’s approval relied on a pivotal clinical study of diluted Radiesse for moderate to severe décolleté wrinkles. The study was prospective, multicenter, randomized, evaluator-blind, and parallel-group, with 152 patients across 9 investigational sites. Patients were treated between December 10, 2021, and January 12, 2023.
Participants were randomized 3:1. The treatment group included 116 patients, while 36 patients entered an untreated control group before delayed treatment. The treatment schedule involved 3 sessions at Day 1, Week 6, and Week 12, with an optional retreatment at Week 36 for the initial treatment group.
The main endpoint looked at improvement at Week 24 using the Merz Aesthetic Scales for Décolleté Wrinkles at rest. FDA documents describe a responder as someone with at least a 1-point improvement from baseline on the scale.
What The Results Showed
At Week 24, the diluted Radiesse treatment group had a 71.2% responder rate on the wrinkle-at-rest scale, compared with 6.3% in the untreated control group. FDA reviewers described the result as clinically and statistically significant.
The response did not vanish quickly after the primary endpoint. In observed cases, responder rates in the treatment group were 72.6% at Week 24, 65.9% at Week 36, 73.3% at Week 48, and 60.7% at Week 60 after initial injection.
Patient and investigator impressions also supported the scale-based results. At Week 24, 92.6% of treatment-group patients showed some improvement according to investigator Global Aesthetic Improvement Scale scores, while 87.4% self-reported some improvement.
FDA data also show patient satisfaction with appearance, tightened skin, fine lines and wrinkles, and smoothness all above 80% at Week 24.
Safety Details Patients Should Know
The FDA summary reported that 11.7% of treated patients had at least 1 treatment-related treatment-emergent adverse event. The most common were injection site bruising at 5.1%, discoloration at 2.9%, injection site mass at 2.2%, and injection site pain at 2.2%.
Most treatment-related events were mild, none were severe, none were late onset, and all resolved without sequelae.
Patient diaries showed common treatment responses after injection, including redness, bruising, pain or discomfort, lumps or bumps, swelling, discoloration, and itching.
Most diary-reported reactions after the first treatment were mild to moderate, and most resolved within 14 days.
The labeling still carries important warnings. Intravascular injection of soft tissue fillers can cause rare but serious events, including embolization, vessel occlusion, ischemia, infarction, vision impairment, blindness, stroke, skin necrosis, and damage to deeper structures.
Treatment should also be deferred when active inflammation or infection is present near the injection area.
Breast Imaging And Training Requirements
The décolleté location raises an obvious question: could an injectable near the chest interfere with breast imaging? FDA documents describe a retrospective review of mammograms, breast ultrasounds, and breast imaging reports after diluted Radiesse treatment.
In the reviewed participants, the FDA reported no evidence of product visible in breast imaging and no interference on mammogram or ultrasound images collected after décolleté treatment.
The FDA also required a post-approval study focused on radiographic imaging and safety. The listed study is designed to assess whether diluted Radiesse in the décolleté interferes with mammography or breast ultrasound, with 30 female participants aged 40 to 65 and a follow-up lasting 60 weeks.
Training is another key part of the approval. FDA documents say the manufacturer must maintain a device-specific use training program covering décolleté anatomy, vasculature, safe injection technique, and management of potential adverse events. Distribution is limited to providers who complete the required indication-specific training.
Who May Be A Reasonable Candidate
A reasonable candidate is usually an adult bothered by moderate to severe upper chest wrinkles who wants a non-surgical option and can accept gradual improvement.
The pivotal study enrolled women aged 30 to 65 with moderate to severe décolleté wrinkles, although the FDA-approved indication covers patients 22 and older.
FDA labeling says safety has not been established for décolleté use during pregnancy, while breastfeeding, or in patients under 22. It also says safety has not been established in décolleté patients with breast cancer, a history of breast cancer, or a familial history of breast cancer. A typical visit starts with assessment of the central upper chest, photography, consent, and marking of the approved treatment region. FDA directions describe a treatment region of roughly 100 cm², bounded above by the sternoclavicular notch, laterally by the midclavicular line, and below by the superior point of the intermammary cleft. No injections should be made over or including breast tissue. The product is diluted, then placed subdermally using a flexible cannula with a threading or fanning technique. A provider may massage the area for even distribution. Afterward, mild redness, swelling, bruising, or tenderness can occur, especially during the first days after treatment. Still, approval does not mean every patient should receive Radiesse, and it does not replace good clinical judgment. Chest wrinkles can involve pigment, sun damage, laxity, etched lines, or all 4 at once. Some patients may benefit more from resurfacing, IPL, radiofrequency microneedling, sunscreen discipline, or a staged combination plan. Radiesse’s new FDA approval for upper chest wrinkles gives patients and trained providers a clearer path for treating décolleté aging with diluted calcium hydroxylapatite. The strongest data show meaningful wrinkle improvement at 24 weeks, sustained response in many patients beyond a year, and a safety profile dominated by temporary injection-site reactions. The best results will depend on careful selection, proper dilution, trained technique, and honest expectations.
What A Treatment Visit May Involve
What The Approval Means For Aesthetic Medicine
The new Radiesse indication shows how body-focused aesthetics is becoming more evidence-driven. Patients already ask for treatment plans that include the face, neck, hands, and chest as one visual field. FDA-cleared décolleté use gives providers a more defined option for an area where improvised off-label plans were common.Summary
Related Posts: